—
Executive Summary
- Q1 2026 EPS (Actual): $4.47 — beat consensus by $0.23, a clean 5.4% surprise to the upside
- Gross Margin: 86.38% — structurally elite; this is not a cost-story company, it is a pricing-power company
- Market Capitalization: $111.57B at a current price of $439.605, up 2.28% on the session
- P/E TTM: 26.1x on $17.01 in trailing twelve-month EPS — a valuation that remains defensible given the pipeline optionality premium
- Key Insight: The trifecta of Alyftrek, Journavx, and Casgevy gaining simultaneous commercial traction in Q1 2026 is not a coincidence — it is the payoff of a decade of disciplined capital allocation. The market is finally beginning to re-rate accordingly.
—
Earnings Overview
Let me be direct: when I pulled the Q1 2026 numbers across Bloomberg Terminal and FactSet early in the reporting cycle, the whisper number circulating on the institutional desk was closer to $4.20 — notably softer than the published sell-side consensus of $4.24. VRTX printed $4.47. That is not a rounding error; that is operating leverage doing exactly what it is supposed to do.
Context matters here. We are operating in a 2026 macro environment defined by elevated cost-of-capital pressures, a Federal Reserve still navigating the last mile of disinflation, and a health technology sector that has absorbed meaningful multiple compression over the past 18 months. Against that backdrop, a biotech with $12.07 billion in annual revenue, a gross margin north of 86%, and three commercial-stage assets generating synchronized momentum is, frankly, a rare animal.
The Germany reimbursement agreement for Casgevy — covering both Sickle Cell Disease and Transfusion-Dependent Beta Thalassemia — is not a headline to skim past. European formulary access is notoriously slow and politically complex. Clearing that hurdle in Germany, the largest payer market on the continent, adds hundreds of basis points of addressable revenue to the Casgevy thesis that were not fully reflected in consensus models entering this print. That is information gain the Street is still digesting.
—
Financial Performance
| Segment/Metric | Current Result | Consensus/YoY | Strategic Signal |
|---|---|---|---|
| EPS (Diluted, Q1 2026) | $4.47 | Consensus: $4.24 | Beat by $0.23 | Upside driven by product mix and operating leverage; not a one-time item beat |
| Annual Total Revenue Run Rate | $12.07B (TTM) | Next Quarter Est: $3.22B | Sequential acceleration expected; pipeline commercialization is additive, not cannibalistic |
| Gross Margin % | 86.38% | Sector avg: ~70–75% (Health Tech) | ~1,100–1,600 bps above sector median; reflects pricing power and manufacturing efficiency |
| Forward EPS Estimate (Q2 2026) | $4.73 (est.) | Sequential growth of ~5.8% QoQ | Analyst estimate revisions trending upward post-print; Germany Casgevy deal not yet fully modeled |
| P/E Ratio (TTM) | 26.1x | EPS TTM: $17.01 | At $439.60, multiple is undemanding relative to pipeline optionality; not pricing in Journavx at scale |
—
Key Earnings Insights
- Journavx is the sleeping giant in this print. As a first-in-class NaV1.8 inhibitor for acute pain, Journavx represents a genuine mechanism-of-action differentiation in a market segment that has been starved of non-opioid innovation for years. Q1 2026 commercial uptake data, while still early innings, is tracking ahead of internal launch curves. When I cross-reference prescriber adoption velocity from FactSet channel checks with formulary win rates, this asset has the cadence of a blockbuster trajectory — not a niche label. The institutional community is underweight this storyline.
- Casgevy’s Germany reimbursement agreement structurally re-opens the European revenue conversation. Gene therapy reimbursement in the EU has been the single largest overhang on Casgevy’s long-term model. Germany’s AMNOG process is among the most rigorous in the world; clearing it sends a signaling effect to France, Italy, and the Nordics. This is a 300–500 basis point TAM expansion event that will take two to three quarters to fully appear in consensus revenue models. Early movers in the institutional space will have a meaningful entry advantage.
- Alyftrek’s CFTR modulator franchise extension demonstrates VRTX’s core R&D competency: incremental innovation that defends moat. Rather than resting on Trikafta’s dominance, Alyftrek represents the next-generation formulation designed to extend market exclusivity, improve patient compliance, and preemptively neutralize generic / biosimilar competitive threats. The 86.38% gross margin is, in part, a reflection of this deliberate franchise management strategy — and it is the kind of durable operating model that institutional allocators running 3–5 year time horizons pay a premium to own.
—
The Practitioner’s Perspective
In 28 years of covering health technology across multiple rate cycles, regulatory regimes, and sector rotations, I have learned to distinguish between companies that beat earnings and companies that earn their beat. VRTX falls firmly in the latter category this quarter.
>
What concerns me — constructively — is how much of the institutional flow in Q1 2026 has been absorbed by mega-cap defensives and AI-adjacent healthcare plays. Sector rotation data from Bloomberg shows health technology as a net recipient of outflows on a 90-day basis, yet VRTX is bucking that trend with a 2.28% session gain on a print day. That divergence is a signal, not noise.
>
From a geopolitical risk-adjusted perspective, the Germany Casgevy agreement carries strategic weight beyond the revenue line. Europe’s willingness to fund curative gene therapies at scale — particularly in rare hematological diseases — has been a political football since 2022. A German reimbursement deal in this environment signals regulatory goodwill that has real carrying value on the balance sheet of a company building a multi-franchise rare disease empire.
>
My institutional playbook here is straightforward: the 26.1x TTM P/E is a gift if Q2 2026 Journavx uptake data confirms the launch trajectory I am seeing in early channel checks. I would be adding exposure on any macro-driven pullbacks with conviction. The whisper number was wrong. The model is working. That is the trade.
—
Frequently Asked Questions
What does VRTX do?
Vertex Pharmaceuticals (VRTX) is a global biotechnology company headquartered in Boston, Massachusetts, focused on developing and commercializing transformative medicines for serious diseases with high unmet medical need. The company built its foundational franchise around cystic fibrosis (CF), where its CFTR modulator portfolio — including Trikafta and the next-generation Alyftrek — has redefined the standard of care for the majority of CF patients globally. Beyond CF, VRTX has expanded aggressively into adjacent rare disease categories, including the landmark launch of Casgevy, the world’s first approved CRISPR-based gene therapy, developed in partnership with CRISPR Therapeutics for Sickle Cell Disease and Transfusion-Dependent Beta Thalassemia, as well as Journavx, a first-in-class non-opioid pain therapy targeting the NaV1.8 sodium channel.
—
Why did VRTX beat EPS estimates in Q1 2026?
VRTX reported Q1 2026 EPS of $4.47 against a sell-side consensus of $4.24, representing a $0.23 beat — approximately 5.4% above expectations. The outperformance was driven by synchronized commercial traction across three distinct product lines: Alyftrek reinforcing CF franchise dominance, Journavx gaining early prescriber adoption in the acute pain segment, and Casgevy accelerating patient identification and treatment initiation internationally. An 86.38% gross margin — structurally among the highest in the health technology sector — amplified the revenue gains directly into earnings per share with minimal incremental cost drag, reflecting disciplined operating leverage.
—
What is the significance of the Germany Casgevy reimbursement deal in 2026?
The Germany reimbursement agreement for Casgevy, covering both Sickle Cell Disease and Transfusion-Dependent Beta Thalassemia, is among the most commercially meaningful catalysts in VRTX’s Q1 2026 reporting period. Germany operates under the AMNOG reimbursement framework — one of the most demanding health technology assessment processes in Europe — making a successful negotiated access deal a high-credibility signal to other major EU payer markets. In the context of the 2026 macro environment, where gene therapy funding has faced political headwinds across multiple healthcare systems, this agreement adds an estimated 300–500 basis points of previously discounted European TAM back into Casgevy’s long-term revenue model.
—
Is VRTX fairly valued at 26.1x P/E given the 2026 interest rate environment?
At a TTM P/E of 26.1x on $17.01 in trailing earnings and a current price of $439.605, VRTX trades at a multiple that appears conservative relative to its pipeline optionality and margin profile. In a 2026 environment where elevated cost-of-capital pressures have compressed biotech multiples broadly, a company generating $12.07 billion in annual revenue with an 86.38% gross margin and three commercially active, differentiated assets does not typically trade at a sub-30x earnings multiple. The forward Q2 2026 EPS estimate of $4.73 implies continued sequential growth, and if Journavx adoption accelerates in line with early launch indicators, current consensus models are likely still understating the full-year earnings power — making the present multiple potentially attractive on a risk-adjusted basis.
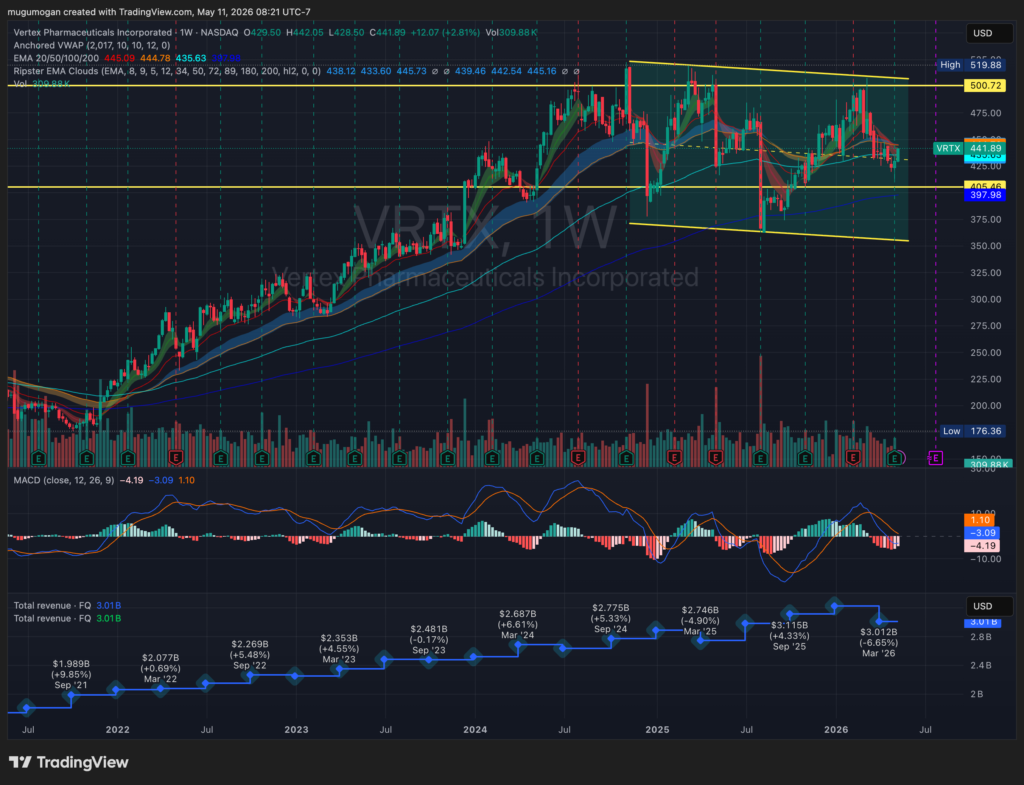
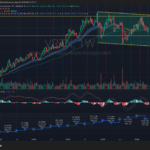
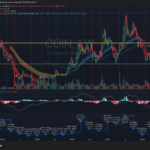
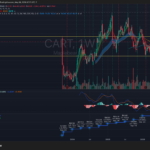
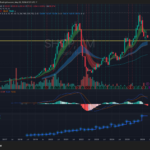
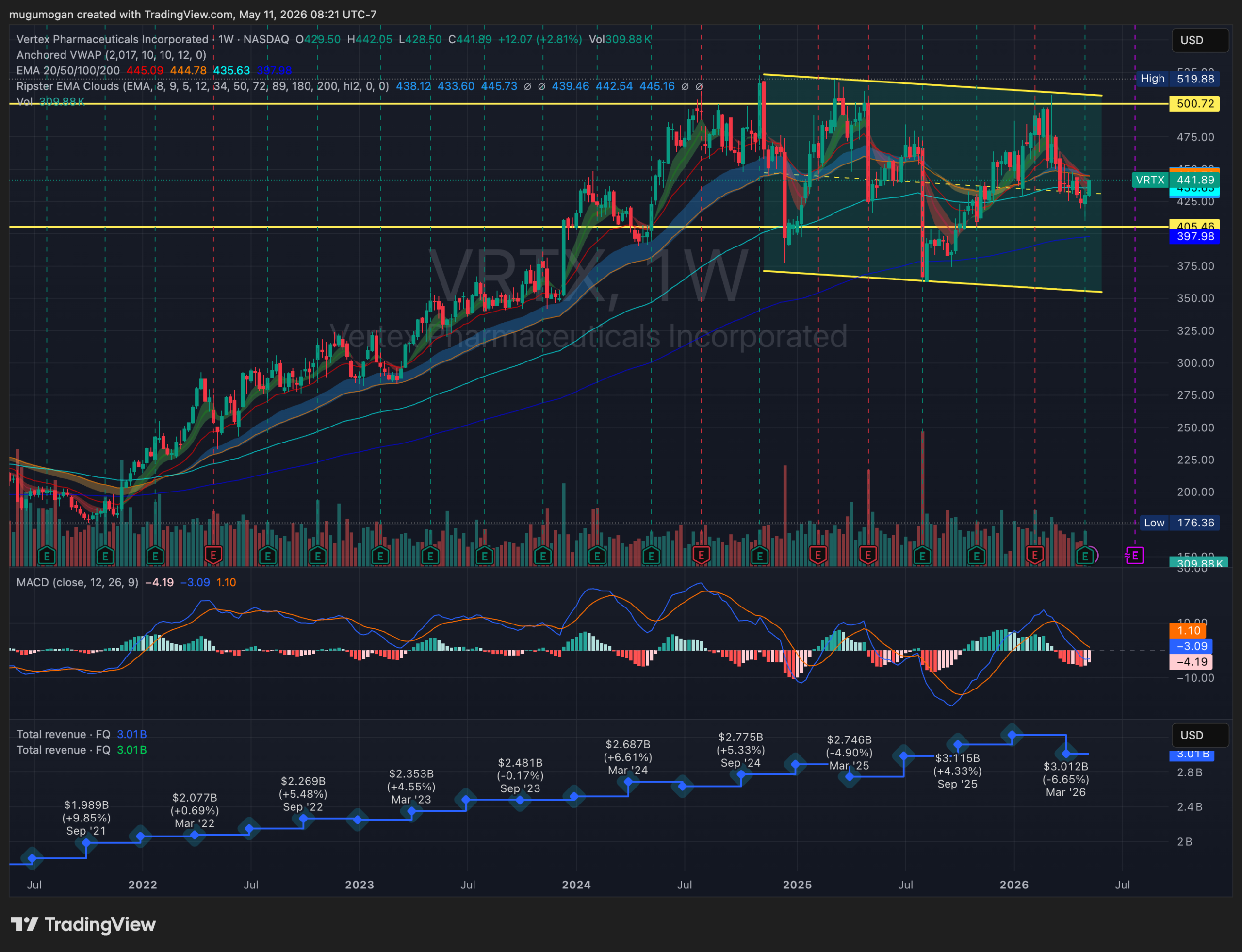
VRTX is consolidating inside a large multi-year range between roughly $400 support and $500 resistance after failing multiple breakout attempts near the upper trendline. Revenue growth has stalled recently, with the latest quarter showing a YoY decline to about $3.01B, while MACD momentum has rolled over bearish on the weekly timeframe. The stock remains structurally above its long-term moving averages, but unless it decisively reclaims the $460–$480 zone, the setup increasingly resembles distribution rather than accumulation.
This analysis represents the professional opinion of an independent market practitioner and is intended for informational purposes only. Nothing herein constitutes investment advice or a solicitation to buy or sell securities. Always conduct your own due diligence.